Methylene Blue Protonation States and pKa Values
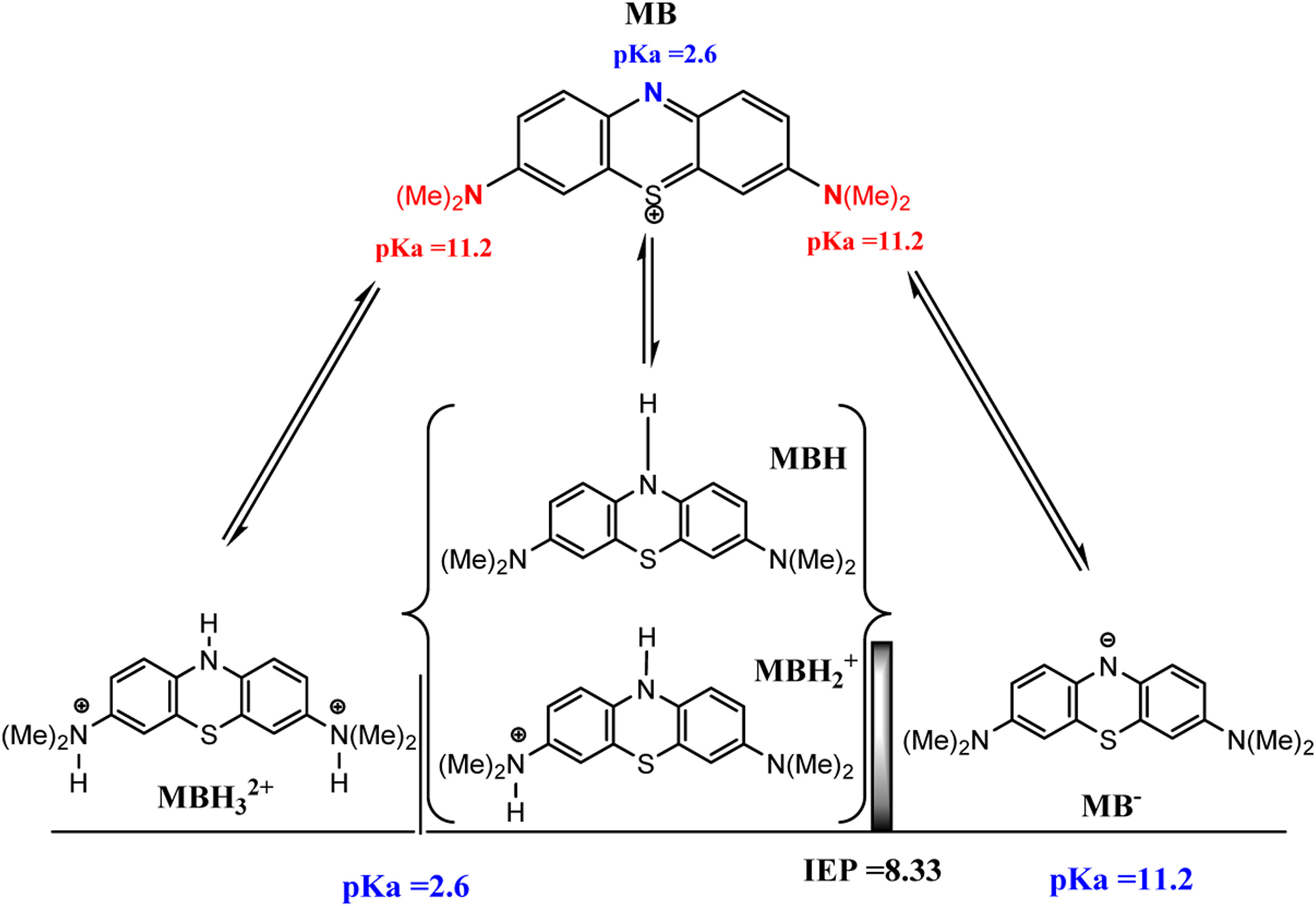
Diagram illustrating the acid-base equilibria of methylene blue (MB), displaying its various protonated and deprotonated forms (MBH₃²⁺, MBH₂⁺, MBH, MB, MB⁻) with associated pKa values of 2.6 and 11.2, and the isoelectric point (IEP) at 8.33.
This educational diagram depicts the acid-base behavior of methylene blue, a thiazine dye. At the top is the cationic methylene blue (MB) structure with pKa = 2.6 labeled near the central nitrogen. Arrows show reversible protonation equilibria leading to multiple species: the triprotonated MBH₃²⁺ (left, pKa = 2.6), the diprotonated MBH₂⁺, the monoprotonated MBH (center), and the deprotonated anionic form MB⁻ (right, pKa = 11.2). Additional pKa = 11.2 values are shown for the dimethylamino groups. A vertical shaded bar in the center marks the isoelectric point (IEP = 8.33), indicating the pH where the net charge is zero. Brackets group related species, and the diagram highlights how nitrogen atoms gain or lose protons, changing the overall charge.